Graphene oxide for Lithium-Sulfur batteries
Jul 08, 2015
At IDTechEx we have invited a number of leading players in the graphene industry to write opinion pieces, reflecting on their products , latest research, market insight, and commercialisation experience. We will be releasing these articles over the next few weeks.
This article is by Mr Jesus de La Fuente, the CEO of Graphenea, a Spanish graphene company. Mr de La Fuente discuss the progress with the use of graphene and graphene oxide in Li Sulphur batteries.
To learn more about the industry, please refer to our report Graphene and 2D Materials: Markets, Technologies, and Opportunities 2015-2025
Rapid development of mobile communication devices, electric vehicles, and other energy-hungry machines detached from landlines is stretching the capabilities of current battery technology. Lithium ion batteries (LIBs) are today's dominant technology due to their excellent cycle stability and good charge/discharge rates. However, the energy density packed in LIBs has reached its peak and is becoming a limiting factor for widespread use of mobile energy consumers. Energy density translates into charging speed, which is highly sought after by consumers.
Potential replacements for LIBs are a hot area of research, with energy density and cost the main gauging parameters. The chart below depicts the state of the art (in blue), with LIB leading current technology with energy density equivalent to 160 km (100 mile) electric vehicle independence. At the theoretical maximum, LIBs could give 200 km (130 miles) of independence to EVs, before the need for recharging. In the order of increasing energy density, battery technologies that are being considered as successors of LIBs are zinc-air, lithium-sulfur (Li-S), and lithium-air.
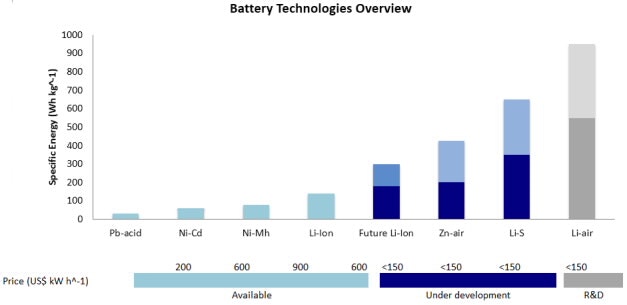
In the mid-term, lithium sulfur (Li-S) will be the most likely successor to LIBs. At the moment, Li-S batteries suffer from poor cycle stability and low rates, withholding a revolution in battery technology. In this respect, graphene oxide (GO) has arisen as an interesting addition to sulfur cathodes, substantially improving the performance of Li-S batteries. Here we review the technology of using GO in Li-S batteries and the advantages that it offers.
Principle of operation of Li-S batteries
Li-S cells typically consist of a lithium anode and a carbon-sulfur cathode, separated by an electrolyte. During discharge, lithium ions from the anode flow through the electrolyte to the cathode, there reacting with sulfur to form polysulfides. Energy is released during the lithium-sulfur reaction. During charging, the reverse occurs, replacing lithium ions in the anode and sulfur in the cathode.
The problem with first-generation Li-S batteries is that the polysulfide drifts away from the cathode and dissolves in the electrolyte, leading to irreversible loss of the active sulfur and a consequent reduction in cycle stability. Cycle stability over at least 1,000 cycles is imperative for commercial batteries. The polysulfides that reach the anode react with the lithium, coating the anode with a passive layer of Li2S, which inhibits anode performance. Graphene-based cathodes emerge as an excellent solution to these challenges and have resulted in tremendous performance improvements.
Graphene-based cathodes
There are numerous ways in which graphene has been applied as a cathode material in Li-S batteries.
Most simply, the conducting graphene electrode can be coated with elemental sulfur, for example by mechanical mixing. A more stable electrode is obtained with a chemical method which starts from GO. Li-S batteries with such cathodes perform better because the graphene electrode conducts better than traditional graphite ones, but the sulfur at the cathode surface is still exposed to the electrolyte and can drift away.
A second approach is to embed sulfur into the graphene electrode, by mixing it with thermally exfoliated graphene. The sulfur then sits in a graphene matrix, which is additionally stabilized by coating with reduced GO (rGO). Coating the matrix with rGO was shown to increase discharge capacity retention by 50%.
Another interesting approach is to coat sulfur microparticles with graphene. Scanning electron micrographs of graphene-coated sulfur particles are shown in Figure X. After the reaction, the graphene "blanket" prevents polysulfides from detaching from the cathode and flowing to the electrolyte. This approach, which also starts from GO, improves Li-S battery cycle stability to 86% after 60 cycles, more than any other method.
Finally, embedding sulfur particles in a graphene hydrogel and directly using synthesized graphene/sulfur paper electrodes have also resulted in Li-S batteries superior to current technology.
Conclusions
Graphene, and in particular graphene oxide, has shown to be a valuable material for solving the hardest challenges presented in lithium-sulfur batteries. Graphenea has continuously expanded its GO product line, now featuring volumes ranging from just 250 ml up to 2,5 l. The company's catalog also holds reduced graphene oxide in powder form and graphene oxide films obtained with vacuum filtration, all ready to be tested in novel cathode designs or mass-produced for the applications described above. Earlier this month, Graphenea was awarded €2.5 million for construction of a production plant, which will expand production capacity by 200 times. The funding was approved by the European Commission, as part of the Horizon 2020 program "SME Instrument". The new production plant should be finished by the end of 2017, accelerating the growth of Graphenea and strengthening its position as a global leader in graphene materials.
References (if needed):
J. Mater. Chem. A, 2015,3, 7241-7247
J. Mater. Chem. A, 2014, 2, 33-47
